8-Step Packaging Compliance Plan
A systematic approach to achieving packaging compliance across PPWR, EPR, and sustainability requirements. Follow these eight steps to transform your packaging portfolio.
By Kevin Kai Wong, Managing Partner at gCurv Technologies
February 22, 2026

Packaging compliance has become complex enough to require a systematic approach. With PPWR, multiple US state EPR programs, recyclability mandates, PCR requirements, and sustainability commitments all demanding attention, companies need a structured methodology to prioritize actions, allocate resources, and track progress. The 8-Step Compliance Plan provides that framework.
The 8-Step Compliance Plan
Step 1: Portfolio Inventory and Data Collection
Before you can improve compliance, you must understand your current state completely.
Actions: - Catalog every packaging SKU with material specifications - Document primary, secondary, and tertiary packaging components - Collect supplier certificates and material declarations - Map packaging to products, channels, and geographies
Key metrics to establish: - Total SKU count by packaging type - Volume/weight by material - Geographic distribution of packaging use
Common gaps: Most companies discover their packaging databases are incomplete. Plan for 4-8 weeks of data collection and validation.
Step 2: Regulatory Mapping
Identify which regulations apply to each packaging SKU based on where products are sold.
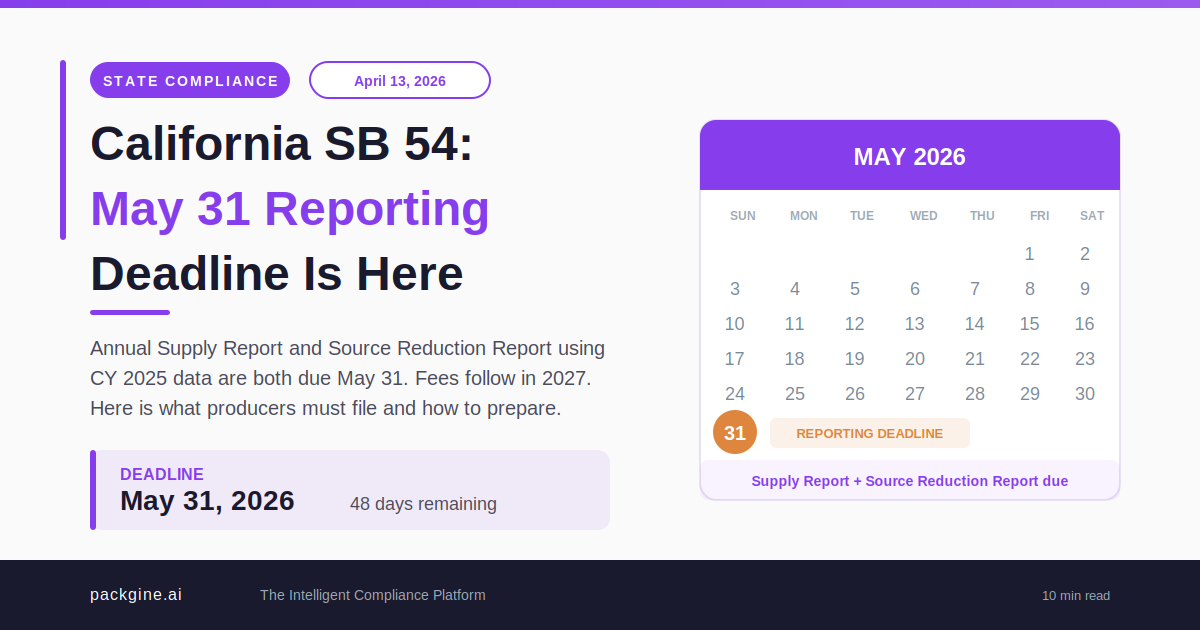
Actions: - Map SKUs to applicable jurisdictions (EU markets, US states) - Identify relevant requirements for each jurisdiction (PPWR, California SB 54, Maine LD 1541, etc.) - Document current and upcoming compliance deadlines - Create a regulatory calendar with key milestones
Output: A regulatory requirements matrix showing which rules apply to which SKUs, with compliance deadlines.
Step 3: Compliance Gap Assessment
Compare current packaging specifications against applicable requirements to identify gaps.
Assessment dimensions: - Recyclability (current grade vs. required grade) - Recycled content (current PCR % vs. mandated minimums) - Substance compliance (PFAS, heavy metals, restricted materials) - Labeling (current labeling vs. required harmonized labels) - EPR registration (registration status in each applicable jurisdiction)
Output: A gap analysis showing compliance status for each SKU/jurisdiction combination, with severity ratings.
Step 4: Risk Prioritization
Not all compliance gaps are equally urgent. Prioritize based on risk and impact.
Prioritization criteria: - Deadline proximity: Issues with imminent deadlines rank highest - Financial impact: Higher-volume SKUs and higher-fee materials warrant more attention - Customer requirements: Retailer or brand customer mandates may create deadlines earlier than regulations - Conversion complexity: Some gaps require simple fixes; others require complete redesigns
Output: A prioritized action list, typically in tiers: Tier 1 (immediate action required), Tier 2 (action within 12 months), Tier 3 (action within 24 months).
Step 5: Solution Identification
For each prioritized gap, identify potential solutions and evaluate alternatives.
Solution types: - Material substitution (e.g., switch from multi-layer to mono-material) - Design modification (e.g., removable sleeve instead of full-body shrink) - Supplier change (e.g., source from supplier with PCR capability) - Format elimination (discontinue non-compliant packaging)
Evaluation criteria: - Compliance effectiveness (does it fully resolve the gap?) - Technical feasibility (can it be manufactured at scale?) - Cost impact (what are material, tooling, and transition costs?) - Timeline (how quickly can it be implemented?)
Output: Recommended solutions for each compliance gap, with cost/benefit analysis.
Step 6: Implementation Planning
Develop detailed project plans for executing recommended solutions.
Planning elements: - Supplier qualification and contracting - Tooling and equipment requirements - Testing and validation protocols - Inventory transition management - Customer communication and approval - Packaging artwork and labeling updates
Timeline considerations: Major packaging changes typically require 12-18 months from decision to full implementation. Plan accordingly relative to compliance deadlines.
Output: Detailed implementation plans with milestones, responsibilities, and resource requirements.
Step 7: Execution and Monitoring
Execute implementation plans while monitoring progress and managing risks.
Execution best practices: - Pilot changes with limited SKUs before broad rollout - Conduct production trials to validate manufacturing performance - Test consumer acceptance where packaging changes are visible - Document compliance evidence (certificates, test reports, specifications)
Monitoring requirements: - Track milestone completion against plan - Monitor costs against budget - Capture lessons learned for future phases - Report progress to leadership
Output: Implemented packaging changes with documented compliance evidence.
Step 8: Continuous Compliance Management
Packaging compliance is ongoing, not one-time. Establish systems for continuous management.
Ongoing requirements: - Monitor regulatory changes and new requirements - Update compliance assessments as regulations evolve - Maintain documentation and certification currency - Conduct periodic portfolio re-assessments - Report compliance metrics to stakeholders
System requirements: - Compliance tracking platform (like Packgine) - Regular review cadence (quarterly minimum) - Clear ownership and accountability - Integration with packaging development processes
Output: Sustainable compliance management capability.
Master Data Management: The Foundation
Underlying all eight steps is master data management, the discipline of maintaining accurate, complete, and current packaging data.
Critical data elements: - Material composition (by layer and component) - Supplier and source information - Regulatory classifications and certifications - Volume and geographic distribution - Cost data (materials, EPR fees, conversion costs)
Data management practices: - Single source of truth for packaging specifications - Defined data ownership and update processes - Integration with PLM and ERP systems - Regular data quality audits
Without robust master data, compliance efforts are built on an unstable foundation.
How Packgine Helps
Packgine is designed to support every step of the 8-Step Compliance Plan.
EPR & PPWR Compliance Automation: Packgine automates Steps 1-3 (inventory, regulatory mapping, gap assessment) and Step 8 (continuous management). Our platform ingests your packaging data, maps it to applicable regulations, identifies gaps, and monitors ongoing compliance, all in real time.
Compliance Cost Estimating: Packgine supports Steps 4-6 (prioritization, solution identification, planning) by modeling the cost and benefit of different compliance approaches. Compare alternatives, project ROI, and build business cases with defensible numbers.
Alternative Product Suggestions: For every compliance gap, Packgine recommends specific solutions, material substitutions, design modifications, supplier alternatives. Our recommendation engine accelerates solution identification (Step 5) and provides implementation guidance.
Implementation Case Studies
Case Study: Mid-Sized Beauty Brand
A mid-sized beauty brand with 350 SKUs selling across the EU and three US states followed the 8-Step Plan over 18 months. Key outcomes included cataloguing 1,200 individual packaging components across all SKUs (Step 1), identifying PPWR recyclability gaps in 42 percent of packaging components (Step 3), converting 85 high-priority SKUs from multi-material to mono-material formats (Steps 5 to 7), and reducing projected annual EPR fees by EUR 180,000 through improved recyclability grades.
The total investment in the compliance programme was approximately EUR 350,000, including staff time, consulting support, packaging redesign, and compliance software. The 18-month payback period demonstrated clear positive ROI.
Case Study: Large CPG Company
A large consumer goods company with 2,500+ SKUs across food, beverage, and household categories implemented the 8-Step Plan as a multi-year programme. Year one focused on Steps 1 to 4 (assessment and prioritisation), establishing a comprehensive packaging database and identifying 600 SKUs requiring remediation action.
Year two addressed Tier 1 priorities (200 SKUs with the highest compliance risk and fee exposure), delivering EUR 1.2 million in annual EPR fee reductions through packaging conversions. Year three continued with Tier 2 priorities while establishing continuous compliance management processes (Step 8) to handle ongoing regulatory changes.
Common Pitfalls and How to Avoid Them
Pitfall 1: Underestimating Data Collection Effort
Most companies significantly underestimate the time and resources required for comprehensive packaging data collection (Step 1). Plan for 8 to 16 weeks of dedicated effort, depending on portfolio complexity, data quality, and the number of suppliers who need to provide information.
Mitigation: Start data collection immediately, assign dedicated resources, and use structured data templates to ensure consistency. Do not wait for perfect data before proceeding to subsequent steps. An 80 percent complete dataset is sufficient for initial gap assessment and prioritisation.
Pitfall 2: Analysing Without Acting
Some companies become stuck in perpetual analysis, continuously refining their gap assessments and prioritisation matrices without proceeding to solution identification and implementation. Analysis is valuable only when it drives action.
Mitigation: Set firm deadlines for completing each step and establish governance mechanisms that require progress reporting. Assign accountability for moving from analysis to implementation.
Pitfall 3: Treating Compliance as a Project Rather Than a Capability
Companies that treat packaging compliance as a time-limited project risk regression after the project team disbands. Regulations continue to evolve, new requirements emerge, and packaging portfolios change constantly.
Mitigation: From the outset, plan for Step 8 (Continuous Compliance Management) as a permanent organisational capability, not a temporary initiative. Invest in sustainable processes, tools, and team structures that persist beyond the initial compliance effort.
Pitfall 4: Siloed Implementation
Packaging compliance requires coordination across sustainability, engineering, procurement, marketing, and finance. When implementation is siloed within a single function, critical dependencies are missed and progress stalls.
Mitigation: Establish cross-functional governance with clear roles and regular coordination meetings. Ensure each function understands its contribution to the compliance plan and has allocated resources accordingly.
Scaling the 8-Step Plan for Multinational Organisations
Multinational companies face the additional challenge of implementing the 8-Step Plan across multiple regions with different regulatory requirements, different packaging portfolios, and different organisational structures.
Centralised Framework, Localised Execution
The most effective approach combines a centralised compliance framework with localised execution. The central team defines the methodology, tools, and standards (Steps 1 to 3), while regional teams execute prioritisation, solution identification, and implementation (Steps 4 to 7) based on local regulatory requirements and market conditions.
Global Data Platform
A global packaging data platform enables consistent assessment methodology across regions, benchmarking and best practice sharing, aggregated reporting to corporate leadership, and identification of cross-regional synergies where a packaging improvement in one market can be replicated in others.
Knowledge Transfer
Regional teams frequently develop solutions to compliance challenges that are applicable in other markets. Establishing formal knowledge transfer mechanisms, including shared solution databases, cross-regional working groups, and periodic capability reviews, accelerates compliance across the organisation and reduces duplicated effort.
Ready to automate your packaging compliance?
See how Packgine manages EPR, PPWR, and sustainability reporting from a single dashboard.